|
5/24/2023 0 Comments Cathode ray experiment conclusions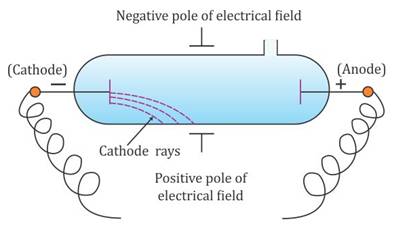
The details were not fully understood until the development of plasma physics in the early 20th century.Ĭrookes tubes evolved from the earlier Geissler tubes invented by the German physicist and glassblower Heinrich Geissler in 1857, experimental tubes which are similar to modern neon tube lights. At higher gas pressures, above 10 −6 atm (0.1 Pa), this creates a glow discharge a pattern of different colored glowing regions in the gas, depending on the pressure in the tube (see diagram). The full details of the action in a Crookes tube are complicated, because it contains a nonequilibrium plasma of positively charged ions, electrons, and neutral atoms which are constantly interacting. The above only describes the motion of the electrons. When the amount of gas in a Crookes tube is a little higher, it produces a pattern of glowing regions of gas called a glow discharge. After striking the wall, the electrons eventually make their way to the anode, flow through the anode wire, the power supply, and back to the cathode. 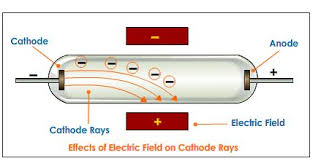
Later on, researchers painted the inside back wall of the tube with a phosphor, a fluorescent chemical such as zinc sulfide, in order to make the glow more visible. The electrons themselves are invisible, but the glow reveals where the beam of electrons strikes the glass. This process, called cathodoluminescence, causes the glass to glow, usually yellow-green. 
When the electrons fall back to their original energy level, they emit light. When they strike atoms in the glass, they knock their orbital electrons into a higher energy level. When they get to the anode end of the tube, they have so much momentum that, although they are attracted to the anode, many fly past it and strike the end wall of the tube. The high voltage accelerates these low-mass particles to a high velocity (about 37,000 miles per second, or 59,000 km/s, about 20 percent of the speed of light, for a typical tube voltage of 10 kV ). These are the cathode rays.Įnough of the air has been removed from the tube that most of the electrons can travel the length of the tube without striking a gas molecule. When they strike it, they knock large numbers of electrons out of the surface of the metal, which in turn are repelled by the cathode and attracted to the anode or positive electrode. All the positive ions are attracted to the cathode or negative electrode. The electrons go on to create more ions and electrons in a chain reaction called a Townsend discharge. The electrons collide with other gas molecules, knocking electrons off them and creating more positive ions. When high voltage is applied to the tube, the electric field accelerates the small number of electrically charged ions and free electrons always present in the gas, created by natural processes like photoionization and radioactivity. The Crookes tubes require a small amount of air in them to function, from about 10 −6 to 5×10 −8 atmosphere (7×10 −4 - 4×10 −5 torr or 0.1-0.006 pascal). 
Instead, electrons are generated by the ionization of the residual air by a high DC voltage (from a few kilovolts to about 100 kilovolts) applied between the cathode and anode electrodes in the tube, usually by an induction coil (a "Ruhmkorff coil"). The term Crookes tube is also used for the first generation, cold cathode X-ray tubes, which evolved from the experimental Crookes tubes and were used until about 1920.Ĭrookes tubes are cold cathode tubes, meaning that they do not have a heated filament in them that releases electrons as the later electronic vacuum tubes usually do. Wilhelm Röntgen discovered X-rays using the Crookes tube in 1895. Crookes tubes are now used only for demonstrating cathode rays. Thomson's 1897 identification of cathode rays as negatively charged particles, which were later named electrons. It was used by Crookes, Johann Hittorf, Julius Plücker, Eugen Goldstein, Heinrich Hertz, Philipp Lenard, Kristian Birkeland and others to discover the properties of cathode rays, culminating in J.J. When a high voltage is applied between the electrodes, cathode rays ( electrons) are projected in straight lines from the cathode. ĭeveloped from the earlier Geissler tube, the Crookes tube consists of a partially evacuated glass bulb of various shapes, with two metal electrodes, the cathode and the anode, one at either end. The anode is the electrode at the bottom.Ī Crookes tube (also Crookes–Hittorf tube) is an early experimental electrical discharge tube, with partial vacuum, invented by English physicist William Crookes and others around 1869-1875, in which cathode rays, streams of electrons, were discovered. Electrons (cathode rays) travel in straight lines from the cathode (left), as shown by the shadow cast by the metal Maltese cross on the fluorescence of the righthand glass wall of the tube.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
